 
When these two conditions are met, the equation is said to be balanced. So now all we need to do is make the left side have 6 H in total. 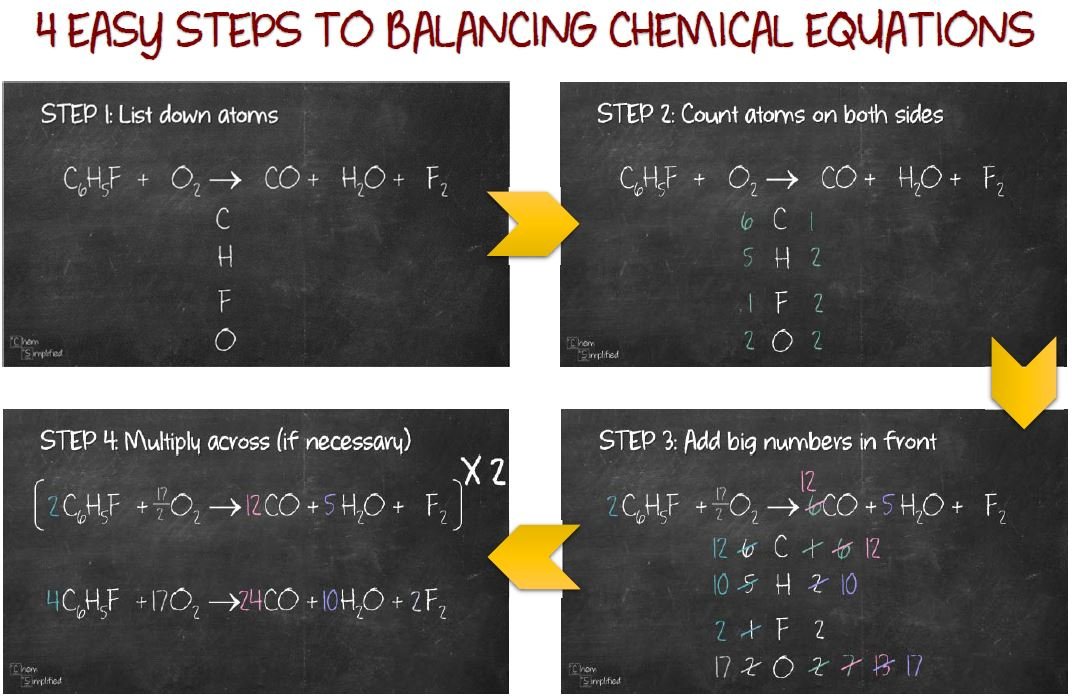
Total the atoms up again: On the left there is 2 N and 2 H still. Also the sum of the charges on one side of the equation must be equal to the sum of the charges on the other side. Theres 2 on the left and 1 on the right, so we need to change the coefficient of NH3 to 2. Use the balanced equation to construct conversion factors in terms of moles. The balanced chemical stoichiometry equations for the water formation reaction is as follows: 2 H 2 O 2 2 H 2 O Step 02: Now you need to write the atomic and molecular masses of each and every atom involved in the reaction: Atomic masses: Hydrogen 1 Oxygen 16 For calculations and steps, tap the atomic mass calculator. This means that a chemical equation must have the same number of atoms of each element on both side of the equation. Balance a chemical equation in terms of moles. The Law of Conservation of Mass states that mass is neither created nor destroyed in an ordinary chemical reaction. If the elements in a chemical formula are properly capitalized, the smart case converter leaves them as you have typed.Ī balanced chemical equation accurately describes the quantities of reactants and products in chemical reactions. Select two compounds above and this calculator will predict whether or not the reaction will occur in water.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
